Nuclear Reprocessing: The Gap Between Promise and Reality
Imagine a technology that could take the most feared byproduct of nuclear power and transform it into fuel: burning the long-lived actinides that make spent fuel a supposed million-year liability down to fission products that decay to background radiation within a few centuries.
All of this while extracting enough energy from the 95,000 metric tons of nuclear waste sitting in pools and dry casks across the United States to power the entire country for 150 years.
That technology has been demonstrated. At Idaho National Laboratory, the Experimental Breeder Reactor II (EBR-2) ran for 30 years on metallic fuel cycled through an attached electrochemical reprocessing facility, closing the loop in exactly the way the vision describes: spent fuel in, fresh fuel out, waste reduced to a centuries-scale rather than geological-scale problem. The physics and engineering has been proven at pilot research scale. It has however failed to scale.
What did scale commercially looks considerably different. La Hague, the world’s largest reprocessing facility, has operated for the better part of a century. France built it not out of a romantic attachment to the closed fuel cycle but out a perceived energy security necessity: Almost no domestic fossil fuels, and, in the postwar decades, uranium supply chains that looked dangerously thin.
French reprocessing works. It has extended the country’s uranium utilization, deferred the need to mine fresh uranium by putting depleted enrichment tails and recovered plutonium back to work as MOX fuel, and given Paris a degree of fuel cycle independence that looks increasingly valuable as the theme of energy security once more rises to prominence with the Iran war and the closure of the Strait of Hormuz.
What French reprocessing does not do is meaningfully reduce the volume of material destined for deep geological disposal. But the La Hague program is not EBR-2. It is a pragmatic industrial process operating within the constraints of thermal reactor physics, and those constraints are severe. The gap between what France actually achieves and what the closed cycle promises is the central story of nuclear reprocessing, and it begins with understanding what is actually sitting in those spent fuel pools.
What Spent Fuel Actually Is
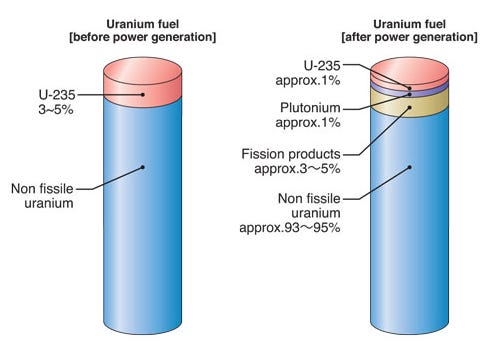
When uranium fuel completes its working life in a reactor, it leaves behind a material of remarkable complexity. A fresh fuel assembly loaded into a pressurized water reactor contains uranium enriched to roughly 5% U-235, the fissile isotope that sustains the chain reaction, with the remaining 95% being U-238, which does not fission under thermal neutron bombardment but plays its own important role. Over the course of 18-24 months in the reactor core, several things happen simultaneously.
The uranium-235 is progressively consumed, splitting into fission products: a zoo of mid-weight atoms, many of them intensely radioactive, which accumulate in the fuel and increasingly poison the neutron economy in the reactor. Separately, some of the uranium-238 absorbs neutrons and transmutes into heavier elements. The most significant of these is plutonium-239, which is fissile and begins contributing meaningfully to the reactor’s power output. By the time a fuel assembly is discharged, roughly half of the energy being generated comes from this in-situ produced plutonium rather than from the original uranium-235.
What emerges from the reactor is therefore not simply depleted uranium. It is approximately 95% uranium, mostly uranium-238 with perhaps 1% uranium-235 remaining, around 1% plutonium spread across several isotopes of varying usefulness, and 3 to 4% fission products.
The fission products are what make spent fuel so radiologically hostile: short-lived but intensely radioactive isotopes generating substantial decay heat and penetrating gamma radiation. The barrier to reloading the spent fuel is primarily that the fission products have made the neutronic environment unworkable and the physical handling of the fuel intolerable. Reprocessing is, at its core, the project of separating the still-useful heavy metals from this contaminating matrix.
The Manhattan Project’s Civil Legacy
The motivation for developing that separation capability did not originate in civilian power generation. Plutonium-239’s properties as a weapons material, chiefly its relatively modest critical mass compared to highly enriched uranium and its producibility from abundant uranium-238, made its extraction a military priority from the earliest days of the American atomic program.
The Hanford site in Washington State was separating plutonium for the Fat Man bomb before a single civilian reactor had generated a watt of commercial power. PUREX, the plutonium-uranium redox extraction process that remains the dominant reprocessing technology today, was a product of that program.
The civilian rationale came later, and it was genuinely compelling given what was known at the time. Postwar estimates of global uranium reserves suggested severe scarcity. If nuclear power was to provide a substantial fraction of the world’s energy, a once-through fuel cycle would exhaust available uranium within decades.
Reprocessing and breeding, in which fast neutron reactors would produce more fissile material than they consumed, appeared not merely attractive but necessary. Scientists at the Chicago Metallurgical Laboratory, working through the physics with the tools available to them, were not wrong to conclude that closed fuel cycles were the only path to long-term nuclear viability.
What they could not know was that uranium would prove far more abundant than early surveys suggested. As exploration intensified, reserves expanded dramatically, and the economics of fresh fuel remained stubbornly favorable relative to the cost of recycling.
When Jimmy Carter banned commercial reprocessing in the United States in 1977, framing it as a nonproliferation measure, the ban was already pointing at a technology that market forces had largely sidelined.
The commercial record made this plain. West Valley in New York, the only U.S. reprocessing plant that ever operated, ran from 1966 to 1972 before shutting down; the estimated cost of upgrades needed to meet newer regulatory requirements rose above $600 million, making restart uneconomic.
Barnwell in South Carolina was partially constructed but never operated: the 1977 policy reversal interrupted construction, and a subsequent GAO review found little commercial interest in completing it given a weak nuclear outlook, unresolved federal policy, and high costs.
The GE Morris plant in Illinois never operated as a reprocessing facility at all; cold testing revealed the technology was more complex than anticipated, the plant would have required extensive modification, and it became a spent-fuel storage site instead. When Ronald Reagan reversed Carter's ban in 1981, the commercial response was revealing: nothing happened. No reprocessing industry emerged. The economics had already delivered the verdict that a rowdy policy debate had obscured.
Inside La Hague
France is the exception that proves the rule, and La Hague is where that exception takes physical form. The largest reprocessing facility in the world, handling around 1700 metric tons of spent fuel per year (equivalent to roughly 17-20 full core loads) at peak operation, La Hague has been running in various forms since the late 1950s. Understanding what it actually does, step by step, is the only way to understand what commercial reprocessing can and cannot accomplish.
Spent fuel arrives in massive shielded transport casks, each containing perhaps a dozen fuel assemblies and weighing enough to require specialized rail or road transport. The assemblies must sit in cooling pools for months before processing begins, allowing the most intensely radioactive short-lived fission products to decay and reducing the thermal load to manageable levels. This waiting period involves a tradeoff that will become important later: the longer the fuel cools, the more certain isotopes decay into daughter products that complicate downstream handling.
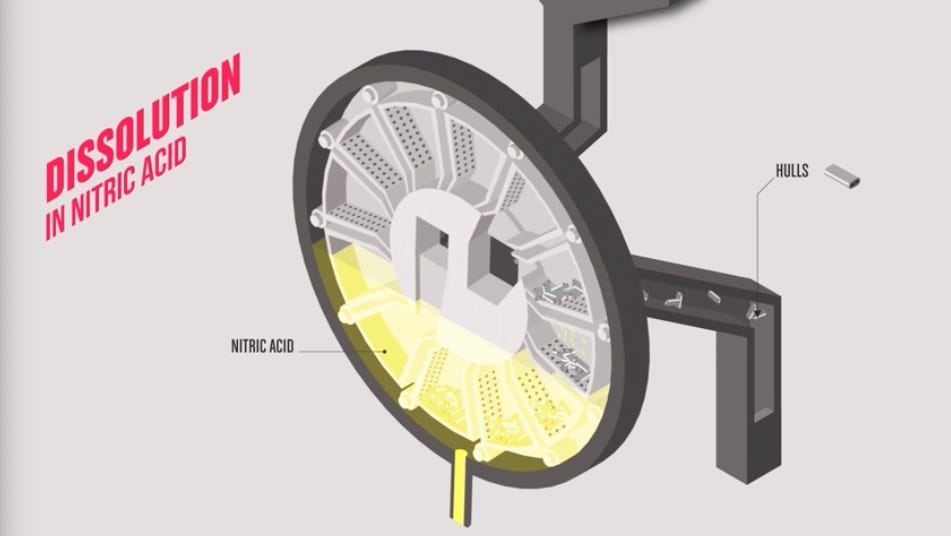
When processing begins, the fuel assemblies are fed into a guillotine chopper: a hydraulically powered blade that slices the entire assembly, zircaloy cladding, ceramic fuel pellets and all, into segments roughly three centimeters long. This is the moment when cladding that has maintained a hermetic seal through 4.5 years of neutron bombardment releases in a single instant the fission gases that have been accumulating inside under pressure. Krypton, xenon, iodine and other volatile species must be captured and filtered rather than vented to atmosphere, and they constitute the first gaseous waste stream the process generates.
The chopped fuel segments then drop into boiling nitric acid. The acid dissolves the uranium dioxide ceramic and releases the heavy metals into solution while leaving the zircaloy cladding hulls behind as a solid residue. Those hulls, too contaminated by neutron activation and fission product migration to recycle economically, are compacted and stored as an intermediate-level waste stream.
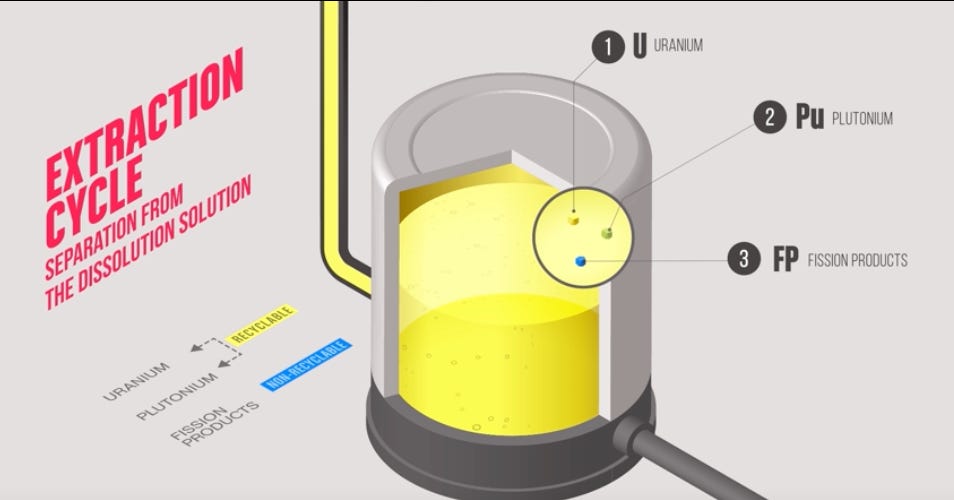
What remains after the acid dissolution is a solution containing uranium, plutonium, fission products, and minor actinides all dissolved together. This solution is then contacted with tributyl phosphate dissolved in a kerosene like hydrocarbon solvent. Here is where the chemistry gets elegant: uranium and plutonium, unlike the fission products, preferentially migrate into the organic phase. By manipulating the oxidation states of uranium and plutonium relative to each other, exploiting the fact that they behave differently under different redox conditions, the process separates them into distinct streams with around 99.5 percent recovery efficiency. The fission products, stripped of the heavy metals they were mixed with, remain in the aqueous phase with almost no cross contamination.
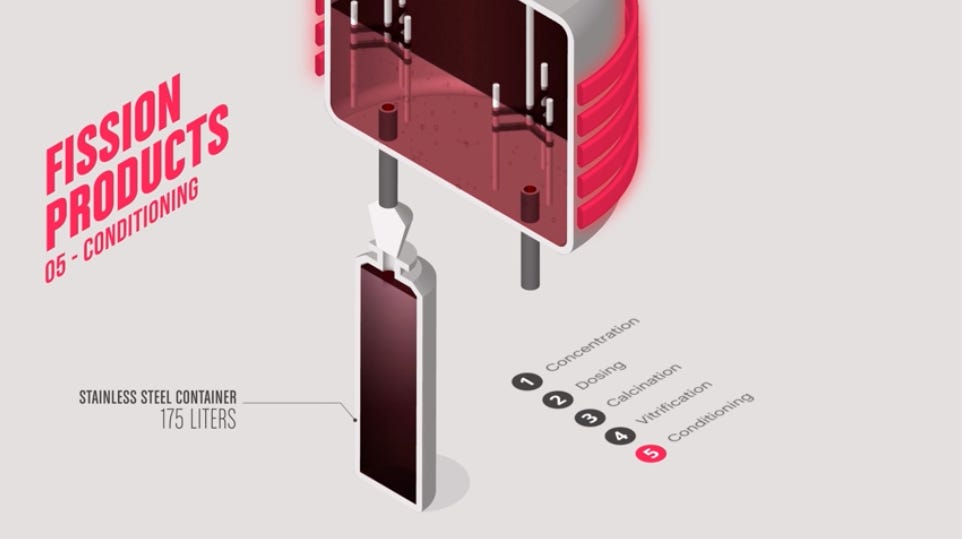
Those fission products are then concentrated, calcined, and incorporated into borosilicate glass, poured into steel cylinders and stored at La Hague. If the fuel came from a foreign utility, France is contractually and legally obligated to return these cylinders to the country of origin, which has created its own geopolitical complications over the years with Japan and Germany. The uranium stream and plutonium stream depart the process separately, each carrying their own challenges.
What the Process Actually Reduces
It is worth pausing on a claim the reprocessing narrative tends to treat as settled: that PUREX substantially reduces the volume of waste requiring deep geological disposal.
The standard figure, repeated by the World Nuclear Association and the IAEA alike, is an 80 to 85 percent reduction in high-level waste volume compared to a once-through cycle. That number is technically defensible in one narrow sense. It compares the volume of vitrified fission product glass canisters against the volume of intact spent fuel assemblies, and since uranium makes up roughly 94 percent of spent fuel by mass, removing it does dramatically shrink the pile of material formally classified as high-level waste. But the comparison omits something important.
In a once-through fuel cycle, the depleted uranium tails from enrichment are a low-level waste problem at most. They are mildly radioactive, carry no fission products, and do not require geological disposal.
When France takes that material, mixes it with separated plutonium to fabricate it into MOX fuel, and irradiates it in a reactor, the depleted uranium that would never have required a deep repository has been converted into spent MOX fuel that under current conventions does.
In addition spent MOX fuel is several times more radioactive than equivalent spent LEU fuel, and its decay heat declines more slowly, meaning it occupies more effective repository space per assembly than the fuel it replaced.
Reprocessing generates multiple additional waste streams that intact direct disposal does not: compacted zircaloy hulls and hardware from fuel disassembly, low- and intermediate-level liquid wastes from the PUREX chemistry, contaminated resins and solvents, and solidified residues from effluent treatment. Each of these streams requires its own handling, conditioning, and disposal pathway.
The total volume of material requiring regulated disposal across all these categories exceeds what would have resulted from simply burying the original spent fuel assemblies whole. Reprocessing does not reduce the waste problem so much as it disaggregates and redistributes it across a more complex and costly management chain.
Standard PUREX also leaves the minor actinides, americium, curium, and neptunium, in the fission product waste stream rather than recovering them. These are the isotopes with very long half lives responsible for most of the radioactivity in the hundred-thousand-year timeframe that regulators believe make deep geological repositories a civilizational-scale engineering necessity.
What reprocessing in a thermal reactor cycle does avoid is one fresh mining cycle and one batch of enrichment tails: a real but narrow benefit, achieved at significantly greater expense and effort.
The Isotopic Trap
The plutonium stream that emerges from PUREX is not weapons-grade material, and understanding why illuminates one of the central constraints on the entire reprocessing enterprise.
Plutonium-239, the isotope produced when uranium-238 absorbs a single neutron, is fissile and desirable both for weapons and for fuel. But uranium-238 does not stop absorbing neutrons after producing plutonium-239. Successive captures produce plutonium-240, which is not thermally fissile and constitutes a neutronic burden; plutonium-241, which is fissile; and plutonium-242, which is not. The longer fuel remains in a thermal reactor, accumulating burn-up, the more the plutonium inventory shifts toward these higher isotopes.
A reactor optimized for weapons-grade plutonium production would discharge fuel after only a few weeks, with burn-up measured in single-digit gigawatt-days per tonne. A commercial power reactor runs its fuel to fifty or sixty gigawatt-days per tonne, producing plutonium that is substantially degraded from a fissile quality standpoint.
This isotopic degradation compounds with each reprocessing cycle. In a thermal reactor, plutonium-239 and plutonium-241 are consumed preferentially during irradiation while plutonium-240 and plutonium-242 accumulate.
After one pass through a MOX fabrication and irradiation cycle, the recovered plutonium contains a significantly higher fraction of non-fissile even numbered non-fissile isotopes. After a second pass, the material is approaching the point of limited utility in a thermal neutron spectrum. The twice-through-then-out cycle that France actually operates is not a policy choice made for lack of ambition; it reflects the physics of what thermal reactors do to plutonium.
The uranium stream carries its own complications. Reprocessed uranium (RepU) is not natural uranium. It contains uranium-236, a neutron absorber that reduces reactivity and uranium-232 which relatively quickly decays through a chain that produces thallium-208, an energetic gamma emitter that makes the material progressively more difficult to handle as it ages. To put it coarsely, instead of aging into a fine wine, reprocessed uranium deteriorates into mouldy grapes. Fabricating it into fresh fuel requires higher enrichment levels and handling radioactive feedstock on production lines designed for essentially inert natural uranium powder, demanding remote or shielded operations where direct human contact was previously routine.
At La Hague, the recovered uranium stockpile has grown for decades. France stores it in the hope of eventual use but has not found an economically or practically compelling route to deploying it at scale. It is the program’s largest stranded asset.
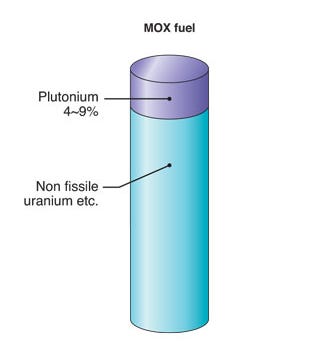
MOX: What the Plutonium Actually Becomes
The separated plutonium does have a destination. Mixed oxide fuel, combining recovered plutonium with depleted uranium tails from the enrichment process, is loaded into 15-20 of France’s fifty-four operating reactors. This is the practical expression of the entire PUREX enterprise, and it functions but with constraints that reveal the limits of the technology in a thermal reactor context.
Fresh LEU fuel contains roughly five percent uranium-235 as its fissile component, with uranium-238 making up the remainder. MOX fuel substitutes plutonium for that fissile loading, with the fissile plutonium fraction typically running between 4 and 8% depending on the isotopic quality of the recovered material and the desired burnup target. The depleted uranium carrier, with its negligible uranium-235 content, is essentially inert from a fission standpoint. From an energy density perspective, the two fuels are broadly comparable in a reactor core
.
The neutronic differences are real, however. Plutonium fission produces a shorter delayed neutron lifetime than uranium-235 fission. Delayed neutrons, the tiny fraction of neutrons emitted seconds after fission rather than instantaneously, are what make thermal reactors controllable on human timescales. A reactor core with a higher plutonium fraction responds more rapidly to reactivity perturbations, tightening the margins that operators and control systems manage.
France limits MOX loading to around 30% of any given core not because higher fractions are physically impossible, but because they would require operators trained and equipped for a reactor that behaves differently from every simulator they have used.
The AP1000 and EPR were designed with full MOX capability in mind, incorporating the additional control authority that a 100 percent MOX core demands, but that capability has not been commercially exercised.
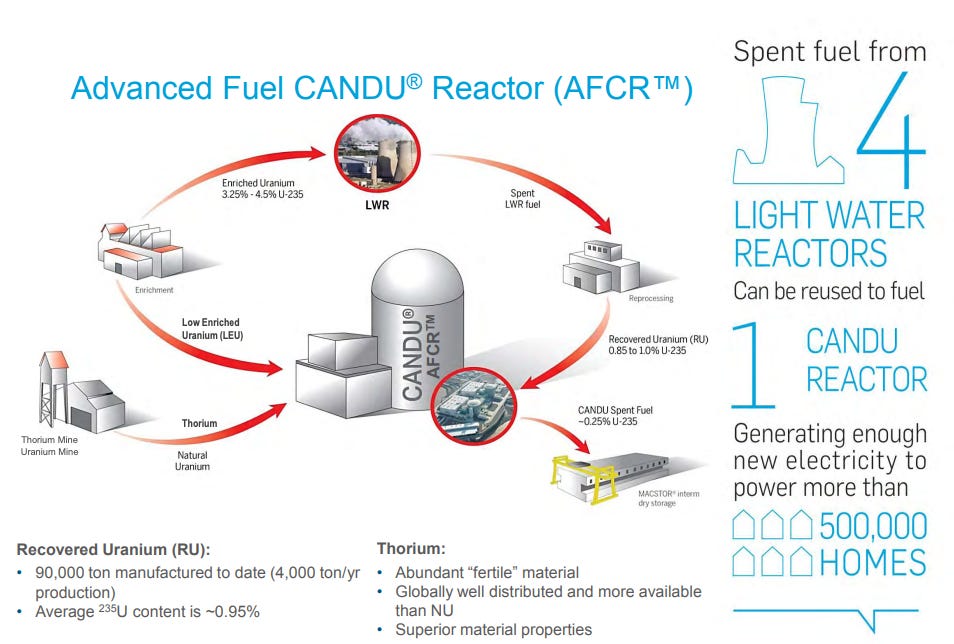
A CANDU solution to the RepU problem? Natural Uranium Equivalent (NUE)
The accumulating stockpile of reprocessed uranium (RepU) at La Hague poses a difficult challenge. CANDU reactors, which run on natural uranium and use heavy water as both moderator and coolant, operate with such a thermally efficient neutron economy that they can sustain criticality on fuel enriched well below the threshold required for light water reactors. RepU, with its residual uranium-235 content of around one percent, sits above the natural uranium threshold and looks, at first glance, like a natural feedstock for a heavy water reactor. The advanced fuel CANDU reactor concept, pursued through a joint venture between Candu Energy Inc. and China National Nuclear Corporation attracted enough political weight to bring the Chinese Premier Li Keqiang and Justin Trudeau to the same signing table.
The concept has genuine logic. Canada possesses the reactor technology capable of using the material. China, which is expanding its commercial reprocessing capability will soon possess RepU in quantities that would otherwise sit indefinitely in storage. This RepU could be fashioned into Natural Uranium Equivalent fuel (NUE) by downblending it slightly with depleted uranium from enrichment tails to be used in CANDU reactors. Both parties saw the potential benefits. What the demonstration program revealed was how much difficulty lies between an appealing concept and a functioning fuel fabrication process.
The small-scale fabrication of the 24 NUE bundles manufactured for the demonstration irradiation was more difficult than foreseen. Specifically selected batches of depleted uranium were needed to match the batches of recycled uranium to achieve an equivalent weight percent uranium-235 to fabricate the pellets.
There were problems with dry blending the depleted uranium and recycled uranium for sintering the pellets, because the grain sizes of the uranium dioxide powder had very large variations. To overcome this issue, the material had to be dissolved in acid and put through a conversion process back to uranium dioxide powder, essentially reintroducing a wet chemistry step into a fuel fabrication process whose simplicity was part of the CANDU value proposition. The challenges with large-scale production were not resolved. NUE has not been used further in the Qinshan reactors.
The reasons reach beyond fabrication difficulty. A CANDU fueling machine loads individual bundles into pressure tubes that run horizontally through the reactor, with fresh and spent bundles moving through each channel during power operation.
A core loaded with RepU bundles of variable enrichment introduces neutronic asymmetries that the standard operating model was not designed to manage. The uranium-232 decay chain imposes a time pressure on using the material before it becomes too radioactive for conventional handling. Lastly the reprocessed uranium’s isotopic composition, variable across different source batches and different cooling histories, resists the kind of precise characterization that stable reactor operation demands.
Each obstacle individually might be surmountable; collectively, they have proven sufficient to stall the program.
The Fast Reactor Promise and Pyroprocessing
Everything described so far takes place in the thermal reactor world, where neutrons are moderated to low energies and the isotopic limitations of recycled plutonium become progressively more binding. The genuine closure of the nuclear fuel cycle, the version where ninety-five thousand metric tons of American spent fuel might theoretically power the country for a century and a half, requires a different neutron spectrum entirely.
Fast reactors, in which unmoderated neutrons sustain the chain reaction, can fission isotopes that thermal reactors cannot. Plutonium-240, the accumulating burden of every thermal reprocessing cycle, fissions readily under fast neutron bombardment. So do the minor actinides: americium, curium, neptunium, that constitute the long-lived hazard in spent fuel.
A fast reactor with a closed fuel cycle does not merely extend uranium utilization; it progressively burns down the transuranic inventory that makes geological disposal a deep-time engineering problem. The fission products that remain are genuinely short-lived on geological timescales, decaying to background levels within a few centuries rather than hundreds of thousands of years.
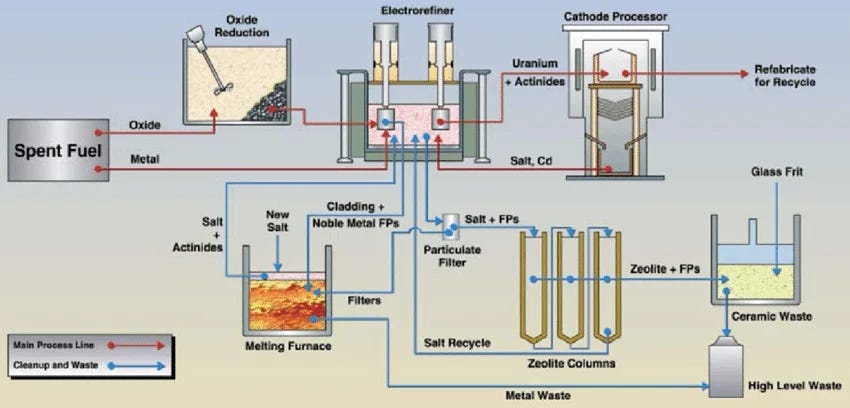
The reprocessing technology best suited to this vision is pyroprocessing rather than PUREX. Instead of dissolving spent fuel in nitric acid and manipulating liquid-liquid extraction chemistry, pyroprocessing dissolves metallic fuel in a molten chloride salt and applies an electrical potential across electrodes.
Heavy metals migrate electrochemically, leaving fission products behind. The process does not separate plutonium from uranium or other actinides for that matter. They all migrate together, which is simultaneously a proliferation advantage and an operational feature for fast reactor fuel cycles that want to keep them mixed. EBR-2 at Idaho National Laboratory ran for decades on metallic fuel with an attached pyroprocessing facility, demonstrating the integrated concept at a small research scale.
The gap between that demonstration and commercial deployment encompasses the entire history of fast reactor development. Despite heroic efforts by the USA, Japan, France, India, Russia and China, no fast reactor has achieved the reliability and economics of the mature light water reactor fleet. In fact the only fast reactors currently operating at all are two units in Russia, one in China and one in India, the PFBR, which achieved its 1st criticality on April 6th of this year after more than 20 years of construction.
The fuel required for pyroprocessing, metallic uranium-plutonium-zirconium alloy, is used in no commercial power plant anywhere in the world including those Russian, Chinese and Indian fast reactors all of which use MOX.
Pyroprocessing has been demonstrated in batches measured in kilograms, not the hundreds of tonnes per year that a meaningful contribution to spent fuel disposition would require. The economic structure of building new pyroprocessing capacity is significantly impacted by capital cost recovery. If built the early decades of operation will impose a heavy fixed-cost burden regardless of throughput. La Hague can offer relatively competitive reprocessing because its capital costs are long since written down; a new pyroprocessing facility would need to recover its full construction cost against a fuel market priced on the marginal economics of existing enrichment capacity. Oklo’s peak valuation of $21 billion reflects the market’s appetite for the narrative more than its assessment of the near-term industrial reality.
The Permanent Condition
America’s 95,000 metric tons of spent fuel await disposal. The physics and processes that make them potentially valuable is proven and has not changed. Uranium-238, transmuted to plutonium and cycled through fast reactors with pyroprocessing, could in principle extend the energy yield from existing fuel stocks by factors that dwarf what PUREX and MOX achieve in practice.
What the history of reprocessing demonstrates however, is that the distance between physical principle and economic reality is vast. As with many compelling solutions to vexatious problems the pilot program exists. Scaling and making a process commercially viable remain the core challenge determined in this case by isotopic decay chains, tight fabrication tolerances, material accounting protocols, and high capital costs amortized over small batches of fuel for reactors that have failed to become commercially viable.
France has run the world’s most ambitious civilian reprocessing program for the better part of a century and extended its uranium utilization by roughly 10% at the fleet level. This is a meaningful achievement but far from being transformative.
Nuclear energy lives permanently in this tension. It must bridge the gap between compelling narratives and the grinding cost and difficulty of the engineering.
Reprocessing has arrived at the destination the physics and economics permitted, which turned out to be considerably short of where the dream was pointing.










Great overview of the technical and economic challenges of reprocessing, but there is particular error I think it necessary to point out:
"The fuel required for pyroprocessing, metallic uranium-plutonium-zirconium alloy, is used in no commercial power plant anywhere in the world including those Russian, Chinese and Indian fast reactors all of which use MOX."
This is not correct; in fact, the very process diagram you use illustrates the head-end oxide reduction process which would be a necessary pre-conditioning step for an oxide-based fuel input to pyroprocessing. Yes, the fuel needs to be a metallic form before electrochemical separations, but this is the whole point of the oxide reduction process.
It's plenty fair to point to the lack of scale-up demonstrated for electrochemical processing, especially as in its current form, it has only been demonstrated as a batch process rather than a continuous process like aqueous processes such as PUREX. But it's not impossible to adapt it to an oxide-based fuel cycle.
Chris,
Under current policy, La Hague does reduce the volume of waste by about a factor of five. Of course, the depleted tails should not require deep geological disposal. But they are RADIOACTIVE. The reason the tails don't need deep disposal is that they produce nearly no gamma radiation, The alpha and beta radiation that uranium-238 and eventually its daughters has little or no penetrating power. This stuff must be swallowed to do any harm, and even then your main problem would be uranium's chemical toxicity. Uranium's biological half-life in humans is about two weeks, next to none of the swallowed U-238 decays inside our bodies.
But the same thing is true of all the uranium and transuranics. For practical purposes, all the gamma emitters are gone in about 500 years, after which ALL the spent fuel can be land filled. By your argument, La Hague reduces the amount of spent fuel that must be sent to geological disposal not at all, because none of it does. Pls check out
https://gordianknotbook.com/download/nuclear-waste-a-tale-of-two-particles/
La Hague proved reprocessing does scale. The reason why we don't see the multi-order of magnitude reduction in mined uranium that was promised by breeders is we don't have any breeders. (Actually, the Russians have a couple, the Chinese have one, and the Indians are about to have one. But at current uranium prices, the economics are still dubious.)
What are the isotopes that complicate reprocessing that show up later in the decay process? This is an honest question. The gamma emitters decay relatively quickly if your idea of quickly is a handful of centuries. After about 500 or so years, the material can be "contact handled" by DOT rules, meaning no shielding is required. That should make processing far easier.
There some some new decay products from spontaneous fission of curium but all the curium is gone in about 200 years. I honestly don't know what those isotopes are.